
The facile, rapid, and complete reductive dissolution of CeO2 and ceria-based oxides, known to be highly refractive materials towards dissolution, is reported for the first time in very mild conditions.
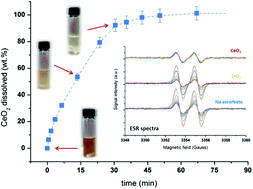
For Pt/CeO2 samples, ceria dissolution goes with Pt aggregation which can be easily recovered from the solution after processing due to its high stability in this medium. This eco-friendly and efficient method appears very promising for catalyst recycling in comparison to conventional procedures. After rigorous characterization of the investigated oxides (BET, SEM-EDX, XRD, TGA), dissolution progress is evaluated with ICP-OES and SEM measurements. The dissolution mechanism is then investigated with UV-Vis, ATR-FTIR and ESR spectroscopies and a general dissolution mechanism is proposed.
Cliquez ici pour accéder à l'article
X. Beaudoux, M. Virot, T. Chave, G. Durand, G. Letdurcq and S. I. Nikitenko. Green Chem. 18 3656 (2016)