
Publication in Catalysts
Team LSFCRecent studies conducted by the ICSM sonochemistry group have demonstrated that increasing the temperature significantly enhances the photocatalytic H2 production from aqueous solutions of 1-BuOH, 2-BuOH, and t-BuOH on the Ti@TiO2 core-shell photocatalyst. The rate of H2 production follows the order 1-BuOH >> 2-BuOH> t-BuOH in the entire temperature range studied (35 – 69°C). In systems with 1-BuOH and 2-BuOH, hydrogen was the only gaseous product observed in the exit carrier gas. In contrast, a mixture of H2, CH4, and C2H6 was detected for t-BuOH, indicating C-C bond cleavage with this isomer during photocatalytic degradation. The apparent activation energies with the 1-BuOH / 2-BuOH isomers (20 - 21 kJ mol–1) were found to be larger than for t-BuOH (13 kJ mol–1). The significant difference in thermal response for the 1-BuOH / 2-BuOH and t-BuOH isomers was attributed to the difference in photocatalytic mechanisms for these species.
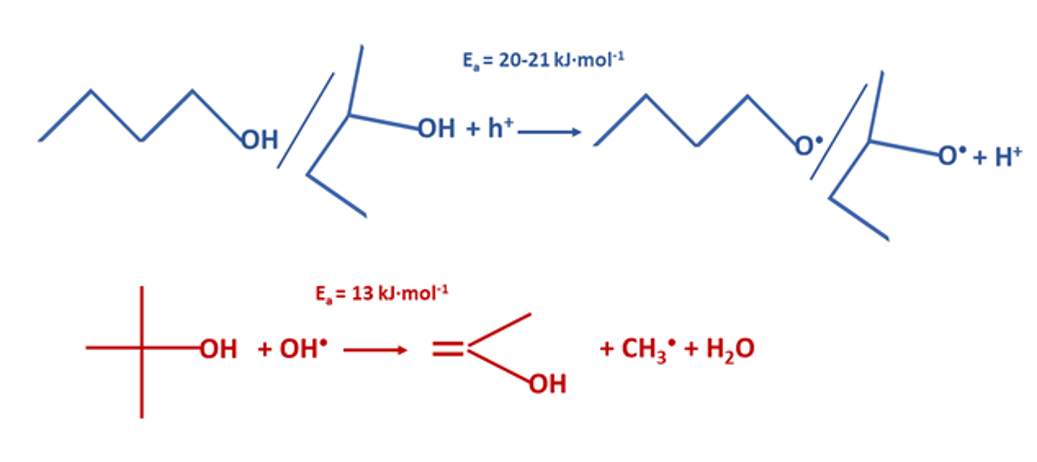
Credit: S. Nikitenko / ICSM
For more information, read the article by Sara El Hakim, Mathéo Bathias, Tony Chave, and Sergey I. Nikitenko in Catalysts 12(12), 1662 (2022). DOI: 10.3390/catal12121662